The original Carnegie Mellon University College of Engineering article by Tara Moore can be read here. ©2021 Carnegie Mellon University
Carnegie Mellon University (CMU) Chemical Engineering and University of Minnesota Chemical Engineering & Materials Science (CEMS) researchers collaboratively used the U.S. Department of Energy’s Advanced Photon Source (APS) in their discovery of a better way to make a new class of soft materials—reducing a process that used to take five months down to three minutes. Their results were published in the journal ACS Macro Letters.
Working with Professors Lynn Walker (CMU) and Mahesh Mahanthappa (UMN), Connor Valentine, a CMU chemical engineering Ph.D. student, and recent UMN CEMS graduate Dr. Ashish Jayaraman study diblock polymers. Diblock polymers are chain-like molecules where one end of the chain is hydrophobic, and the other is hydrophilic. Molecules like this are used in soap because the hydrophobic side grabs onto dirt and oil, but the hydrophilic side keeps the molecules dissolved into the water.
When these molecules are placed in water at high enough concentrations, they begin to form clusters: the hydrophobic parts clump together in the center of the ball in order to avoid water. The hydrophilic sides arrange into a brush-like layer on the outside of the ball, protecting the hydrophobic center.
As you add more polymers into the water, however, they begin to run out of room and pack themselves intelligently and spontaneously. It would be as if you were trying to fit the maximum number of tennis balls in a box—you would carefully stack each layer.
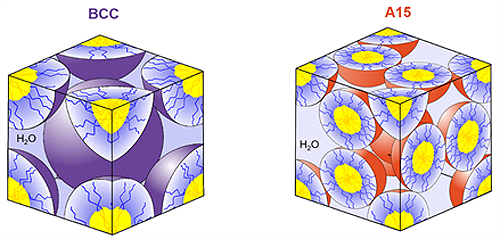
When they form these stacks, they are called crystals, because the organizational patterns will repeat over and over in every direction. Crystalline structures like this are found throughout nature, including in gemstones, metals, and polymeric materials. People have taken advantage of the repeating and consistent spacing to create polymeric membranes for filtering water and gases. There are also exciting potential uses in new soft materials, with applications that include medical implants, adhesives, sustainable food packaging, liquid beauty products, and even condiments.
However, the problem comes in when researchers are trying to make specific crystalline structures. Engineers need to be able to consistently produce crystals with specific arrangements and sizes so that they can achieve the desired material performance at market scale. However, processing issues can arise when they don’t fully understand the forces driving crystal formation. The wrong temperature change, mixing speed, or formulation can cause crystals to suddenly form, degrade, or transition to another crystalline organization. The accompanying change in material properties can jam mixers, ruin equipment, and result in a worthless final product.
In the case of this work, the desired crystalline state can take months to form at room temperature. This can cause huge issues, with companies discovering they have a product with completely different properties after three months—maybe it’s chunky, or it’s become stiff—or perhaps the company just has to wait three months to sell their product because it takes that long to get the gel consistency they desire.
“It’s important for people to understand how these polymer molecules will turn into crystals,” said Valentine. “And that’s not just if they turn into the crystal they want, it’s the rate of it, the speed. Also, are there going to be other crystal phases present? Is every piece of that crystalline material going to be oriented consistently?”
Valentine and Walker worked with collaborators from the University of Minnesota, who discovered that the rate of heating and cooling can produce intermediate crystal structures that last for several months. Valentine’s team built on the initial work of their University of Minnesota collaborators, Ashish Jayaraman and Mahesh K. Mahanthappa, and investigated the impact of shear processing on these crystal structures. Shear processing is a broad term that includes steps like mixing, painting, coating, and shaking—the material is moving. The speed, duration, and direction of shear can really matter for materials like those used in this work.
“Ketchup is a great example about why shear processing affects soft materials because ketchup has a yield stress and thins when you mix or process it,” said Valentine. “If you’re trying to get ketchup out of a glass bottle and it is gel or solid-like, it will not flow. But small taps (on the correct part of the bottle) will get the ketchup to flow very nicely. The shear is changing the microstructure of the ketchup, which then changes the flow properties. It’s important we understand how shear impacts any material we work with in the same way.”
In this work, the authors used an oscillatory shear flow, which involves placing the gel or soft material between two parallel plates—where the top plate can rotate back and forth. Researchers can control the speed and length of the top plate. When Valentine and his team put the diblock polymer crystals into this shear cell, they were able to cause the crystalline phase to change into the equilibrium structure within three minutes. The Minnesota team had previously found this same structural change to take almost five months sitting at room temperature without shear.
“Shear processing can help with the dynamics, the speed, and the rates of structural change, not just the final result, which is something people don’t really think about,” said Valentine. “They often think when you shear these materials, it’s going to change the structure into something different, but that’s not necessarily true.”
The team was able to measure these results by employing small-angle x-ray scattering at the X-ray Science Division Chemical & Materials Science’s 12-ID-B x-ray beamline at the APS, an Office of Science user facility at Argonne National Laboratory. The high-intensity x-ray beams from the APS allowed the research team to to measure the crystal structure in real-time.
Their findings not only showed that the speed increase occurs, but also detailed how to tune the shear parameters to achieve the desired rate of crystal formation. They even found that you can prevent the change from happening entirely if the shear is at very low frequencies with very long cycles of oscillation.
“We were able to show that this shear processing step is just a very controllable way to get the structure you want and how fast you want it,” said Valentine.
See: Connor S. Valentine1, Ashish Jayaraman2, Mahesh K. Mahanthappa2**, and Lynn M. Walker1*, “Shear-Modulated Rates of Phase Transitions in Sphere-Forming Diblock Oligomer Lyotropic Liquid Crystals,” ACS Macro Lett. 10, 538 (2021). DOI: 10.1021/acsmacrolett.1c00154
Author affiliations: 1Carnegie Mellon University, 2University of Minnesota
Correspondence: * lwalker@andrew.cmu.edu, ** maheshkm@umn.edu
M.K.M. and A.J. gratefully acknowledge funding from the National Science Foundation, NSF, (CHE-1807330). C.S.V. and L.M.W. gratefully acknowledge a fellowship from PPG Industries, Inc., and the Dowd Fellows Program at CMU. Preliminary SAXS analyses were performed at the Characterization Facility at the University of Minnesota, which receives partial support from NSF through the MRSEC program (DMR-2011401). This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
The U.S. Department of Energy's APS is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries, and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
