The original Washington University in St. Louis press release by Brandie Jefferson can be read here.
From the splendorous red hues in the Grand Canyon to the mundane rust attacking a neglected bicycle, iron hydroxides are all around us. As a matter of fact, they are just as common as quartz, which is the most widely distributed mineral on the planet. Scientists know that iron hydroxides can capture heavy metals and other toxic materials, and that iron oxides also can be natural semiconductors. While these properties suggest many applications, the full details of how iron hydroxides form on a quartz substrate have been hidden in a “black box” of sorts — until now. Young-Shin Jun, a professor of energy, environmental, and chemical engineering in the McKelvey School of Engineering at Washington University in St. Louis, has devised a way to open that box and observe the moment iron hydroxide forms on quartz with an assist from the U.S. Department of Energy’s Advanced Photon Source (APS). The work was published in Environmental Science & Technology.
“This is telling the story of the birth of iron hydroxide,” Jun said.
When people speak of “formation,” typically they are talking about a substance growing. Before growth, however, there needs to be something to grow. Where does that first bit of iron hydroxide come from?
First, sufficient precursor elements need to be in place. Then the components can come together to form a stable nucleus that will go on to become a tiny solid particle of iron hydroxide, called a nanoscale particulate. The process is called solid nucleation.
Science has a firm grip on the sum of these two processes — nucleation and growth, together known as “precipitation” — and their sum has been used to predict iron hydroxide’s formation behavior. But these predictions have largely omitted separate consideration of nucleation. The results “weren’t accurate enough,” Jun said. “Our work provides an empirical, quantitative description of nucleation, not a computation, so we can provide scientific evidence about this missing link.”
This contribution opens many important possibilities. We can better understand water quality at acid mine drainage sites, reduce membrane fouling and pipeline scale formation, and develop more environmentally friendly superconductor materials.
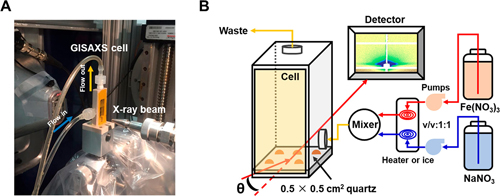
Jun was able to look inside of the black box of precipitation by using x-rays and a novel experimental cell she developed to study environmentally relevant complex systems with plenty of water, ions and substrate material, observing nucleation in real time.
Working at the X-ray Science Division Chemical & Materials Science Group’s 12-ID-B x-ray beamline at the APS, an Office of Science user facility at Argonne National Laboratory, Jun employed the x-ray scattering technique in situ grazing incidence small angle x-ray scattering. The high-brightness x-rays from the APS were directed onto a substrate at a very shallow angle, close to the critical angle that allows total reflection of light; this technique can detect the first appearance of nanometer-size particles on a surface (Fig. 1).
The approach is so novel, Jun said, that when she discusses her lab’s work on nucleation, “People think we are doing computer modeling. But no, we are experimentally examining it at the moment it happens,” she said. “We are experimental observers. I can measure the initial point of nucleation.”
Her empirical method revealed that the general estimates scientists have been using overstate the amount of energy needed for nucleation.
“Iron hydroxide forms much more easily on mineral surfaces than scientists thought, because less energy is needed for nucleation of highly hydrated solids on surfaces,” Jun said.
Furthermore, having a precise value will also help improve reactive transport models — the study of the movement of materials through an environment. For instance, certain materials can sequester toxic metals, keeping them from entering waterways. An updated reactive transport model with more accurate nucleation information will have significant implications for water quality researchers working to better predict and control sources of pollution. “Iron hydroxide is the main sequestration repository for these contaminants,” Jun said, “and knowing their origin is critical to predicting their fate.”
For high-tech manufacturing facilities, having a more precise understanding of how iron oxides or hydroxides form will allow for the more efficient — less wasteful — production of iron-based superconductors.
See: Xuanhao Wu1, Byeongdu Lee2, and Young-Shin Jun1*, “Interfacial and Activation Energies of Environmentally Abundant Heterogeneously Nucleated Iron(III) (Hydr)oxide on Quartz,” Environ. Sci. Technol. 54, 12119 (2020). DOI: 10.1021/acs.est.0c03160
Author affiliations: 1Washington University at St. Louis, 2Argonne National Laboratory
Correspondence: * ysjun@seas.wustl.edu
The authors are grateful for support received from the National Science Foundation Environmental Chemical Science Program (CHE-1214090). This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility, operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
©2021 Washington University in St. Louis
The U.S. Department of Energy's APS is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries, and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
